
The most common vapor is evaporated water - steam or moist. The heat required for changing state as evaporation is referred to as latent heat of evaporation. The heat transferred to a substance when temperature changes is often referred to as sensible heat. the vapor heats above the evaporation temperature - super-heating.the liquid evaporate at the evaporation temperature by changing state from fluid to gas.the liquid heats up to the evaporation temperature.The hot gasses (in the form of steam) have to release energy into the environment in the form of heat to cool to the point that they can form liquid water, meaning that the formation of H 2O is exothermic.When a liquid evaporates its go through a process where This makes sense - H 2 and O 2 are gasses, while H 2O, the product, is a liquid. 
Since the sign is negative, we know that our reaction is exothermic. In our example, our final answer is -13608 J.Beware strongly exothermic reactions - these can sometimes signify a large release of energy, which, if rapid enough, can cause an explosion. The larger the number itself is, the more exo- or endo- thermic the reaction is. On the other hand, if the sign is negative, the reaction is exothermic. If the sign of your final answer for ∆H is positive, the reaction is endothermic. One of the most common reasons that ∆H is calculated for various reactions is to determine whether the reaction is exothermic (loses energy and gives off heat) or endothermic (gains energy and absorbs heat). 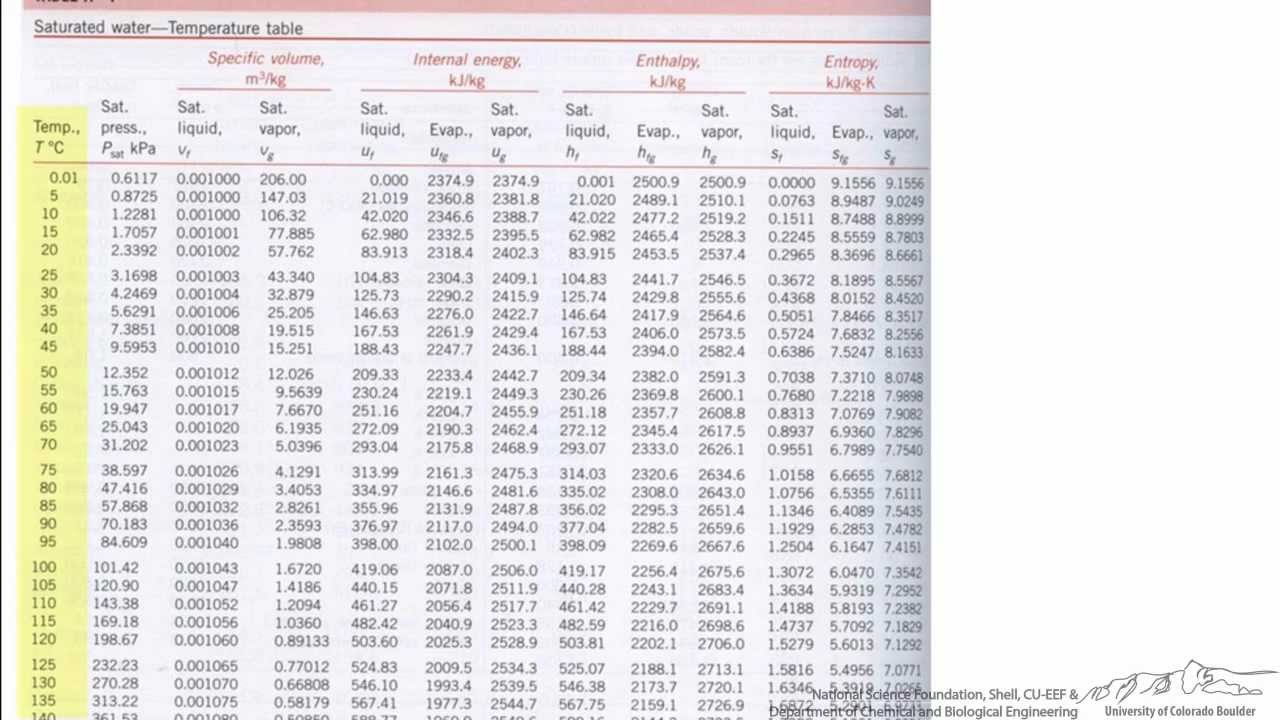 
Determine whether your reaction gains or loses energy.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
